 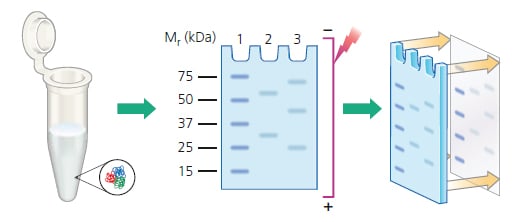
If your protein of interest is in the insoluble fraction (e.g., cell membrane-bound proteins) investigate pretreatment methods to liberate and solubilize it first. For a clean image, samples are centrifuged to remove any solids, in order to load only the soluble fraction. The specific separation method chosen will depend on the aim of the analysis. This is typically achieved by protein electrophoresis, such as sodium dodecyl sulphate–polyacrylamide gel electrophoresis (SDS-PAGE) or native PAGE, which separates proteins based on their molecular weight or charge. Fluorophore Selection.Figure 1: Overview of a western blot protocol.īefore a western blot can be performed, the proteins in the sample must be separated. 
Jackson ImmunoResearch Laboratories Inc.Multiple Labelling for Simultaneous Detection of Several Targets. Choosing the Right Affinity-Purified Secondary Antibody for your Application. Scandinavian Journal of Medicine and Science in Sports. An Overview of Technical Considerations for Western Blotting Applications to Physiological Research. SDS-PAGE and Western Blotting Techniques. We recommend using only solutions of the buffer, without the addition of blocking proteins like powdered milk or BSA.įound Part 7 of our Western blotting guide useful? Get an update when part 8 is available here Wash buffers should be made freshly before use.Ĭaution: Blocking reagents in wash buffersīlocking reagents are sometimes added to wash buffers. Sticky complexes can form and bind to the membrane, causing background Tween-20™ is typically used at 0.05% to 0.1% v/v of the solution.Ĭoncentrations higher than 0.1% can strip the target protein(s) from the membrane and the antibodies bound to them, reducing the signal and accuracy of the blot.Ĭaution: Microbial Growth can create a background signal Detergentsĭetergents such as Triton X-100, SDS, or Tween-20™ are added to prevent aggregation of unbound proteins. It is typically made by combining Tris Base with Tris HCL and sodium chloride to create a solution with a physiological pH of approximately 7.2. TBS is sometimes used as a washing buffer as an alternative to PBS. If the target protein is phosphorylated, or an Alkaline Phosphatase (AP) conjugate is to be used, PBS should be avoided as the phosphate can interact with phosphorylated proteins or quench the AP activity affecting result reliability. PBS solutions can vary in their formulation, but approximate physiological conditions by combining sodium phosphate and sodium chloride to buffer the solution to a pH of 7.5. The washing process is then repeated between each reagent incubation.Ĭhoosing an Appropriate Wash Buffer Phosphate Buffered Saline (PBS).The membrane is incubated in a wash buffer for 5-10 minutes and constantly agitated by using a rocker or platform shaker or suitable blot processor. 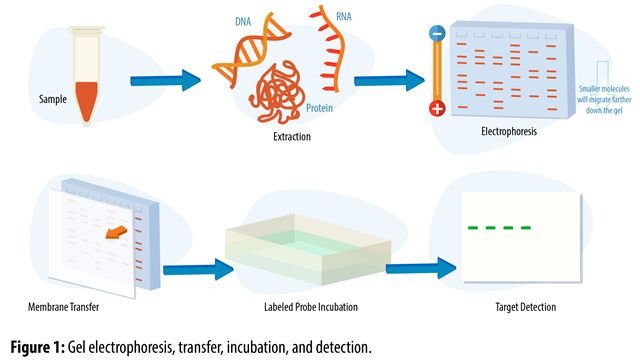
For example, an average 10 cm² blot would need >40 ml of washing buffer, we typically use 100 ml. The blot is washed by placing the membrane in a volume of wash buffer that fully submerges the membrane.Washing is usually performed at room temperature.Washing helps to reduce background signal, improving assay sensitivity by increasing the signal-to-noise ratio and enabling qualitative and quantitative analysis of the protein of interest.The correct washing reagents should be used at the correct concentrations to ensure removal of deleterious agents from the membrane. Washing removes unbound or aggregated proteins present on the blot as well as unbound reagents that may interfere with the detection of the target molecule(s). It is necessary to wash the membrane after blocking and between incubations with the antibodies. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
